Exempt
Sep 26, 2021 14:59:18 #
EmilyD wrote:
It certainly will not be an experimental M-RNA gene manipulation drug concoction that will fix it...
You mean approved v*****es?
Sep 26, 2021 17:51:58 #
moldyoldy wrote:
You mean approved v*****es?
You mean the one that is only in very limited supply right now? The FDA approved the "Comirnaty" drug that was produced by BioNTech....NOT P****r:
P****r did not directly develop its m-RNA shot. It hired a German company in 2018, BioNTech, to do that for them. They subsequently entered into a joint marketing/manufacturing agreement to deliver the resulting product to the world. Thus, BioNTech is not a subsidiary of P****r. It is the BioNTech product that has been approved by the FDA, and its product is labeled "Comirnaty". Because that version has been approved, the liability disclaimer has been lifted, and the company, BioNTech (not P****r), can be held liable for any adverse effects or deaths that may occur. The only problem is, this "Comirnaty" product is available in Europe, but is in very limited supply in the US. What people do not know when they go for the P****r shot, now that they think it's been approved, is that they are being sold a lie!
P****r continues to use the drugs that are still under the EUA authorization only. An EUA authorization is NOT an approval by the FDA. P****r's drugs have NOT been approved by the FDA.
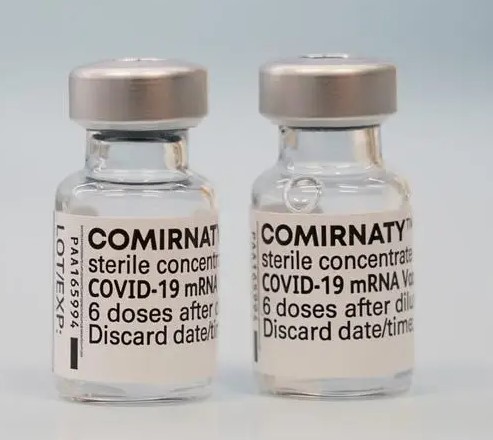
People are not being told that the bottle that is being used to fill the syringe that will be injected into their bodies should have the word "Comirnaty" in large letters on the bottle. If it doesn't, it is NOT the approved version, and that person will not have any recourse (can't sue, and no medical coverage) if they have an a*****e e***t or their families will have no recourse if they die!
The FDA and the mainstream media are promoting this LIE that P****r’s shot has full FDA approval. So are you!!
Here is what people who think they are getting the approved drugs should be looking for on the bottle BEFORE they get the shot....if it doesn't look like this...if it doesn't say "Comirnaty" on it... they aren't getting the approved drugs:
Sep 26, 2021 18:42:52 #
EmilyD wrote:
You mean the one that is only in very limited supp... (show quote)
Common questions
Is Comirnaty the same as P****r v*****e?
The shot has been called the “P****r v*****e” because that's the name of one of the companies that developed it. However, the name change led some people to believe the Food and Drug Administration-approved Comirnaty
And when you have something to promote, you need a brand name. That’s where “Comirnaty” (pronounced koe-mir’-na-tee) comes in. The shot has been called the “P****r v*****e” because that’s the name of one of the companies that developed it.
However, the name change led some people to believe the Food and Drug Administration-approved Comirnaty is a different version of the P****r v*****e — it’s not.
Read more here: https://www.newsobserver.com/news/c****av***s/article253765538.html#storylink=cpy
Sep 26, 2021 19:20:51 #
moldyoldy wrote:
Common questions br Is Comirnaty the same as P****... (show quote)
That is the big lie...the drug that the FDA approved is NOT the P****r drug, but P****r and the media want the public to THINK it is. Anyway, it is about the APPROVAL of the different companies' drugs....not what they named them. Although the name of the one company's drug - "Comirnaty" - will help people distinguish whether they are getting the P****r EUA-only drugs, or the BioNTech's approved "Comirnaty" drugs.
The FDA acknowledges that while P****r has insufficient stocks of the newly licensed Comirnaty v*****e available, there is “a significant amount” of the P****r C***D v*****e — produced under E*******y Use A***********n (EUA) — available for use. And THAT is why P****r doesn't want the public to know the difference between the licensed drugs and the ones that are still under the EUA authorization only.
The FDA decrees that the P****r v*****e under the EUA should remain unlicensed but can be used “interchangeably” with the newly licensed Comirnaty product. How is the public going to know the difference? It's a BIG difference - whether you're covered legally and medically or not!!!
https://www.theburningplatform.com/2021/08/25/2-things-mainstream-media-didnt-tell-you-about-fdas-approval-of-p****r-v*****e/
*************************
Dr. Robert Malone claimed the FDA and media were yet again misleading the public, as they had not approved the P****r shot currently given but rather a P****r/BioNTech version that is not yet available. Malone explained:
“The little trick that they’ve done here, is they’ve issued two separate letters for two separate v*****es. The P****r v*****e, which is what is currently available, is still under e*******y use a***********n and it still has the liability shield …
The product that’s licensed is the BioNTech product, which is substantially similar but not necessarily identical, called Comirnaty, and it’s not yet available. They haven’t started manufacturing it or labeling it for general US public use. And that’s the one the liability waiver will no longer apply to.
So, the one that’s actually licensed is not yet available, and when it does become available it will no longer have the liability shield. In the interim, the one that does have the liability shield is the P****r product and that is what is currently available and it’s still under e*******y use a***********n.”
This would mean that if employers demand that employees get v******ted because there’s now a licensed C***D injection, employees could then demand to actually receive the FDA licensed Comirnaty,18 not the emergency use only19 P****r product that is currently given, and for which you cannot sue for damages in case of v*****e injury.
https://peckford42.wordpress.com/2021/09/14/confusion-over-v*****e-approval-two-key-senior-fda-officials-resign-over-v*****e-decisions/
*************************
The approval letter sent from the FDA to P****r states in footnote 8,
“The licensed v*****e has the same formulation as the EUA-authorized v*****e and the products can be used interchangeably to provide the v******tion series without presenting any safety or effectiveness concerns. The products are legally distinct with certain differences that do not impact safety or effectiveness."
The “certain differences” are not specified, but it is perfectly clear that the two jabs are legally distinct.
https://stateofthenation.co/?p=80552
Sep 26, 2021 20:03:33 #
EmilyD wrote:
That is the big lie...the drug that the FDA approv... (show quote)
The more confusion stoked by the right, the more people will refuse to get the cure.
Phizer makes viagra, the drug has its own name, it would be confusing if every phizer product was called phizer.
Sep 26, 2021 22:09:52 #
moldyoldy wrote:
The more confusion stoked by the right, the more people will refuse to get the cure.
Phizer makes viagra, the drug has its own name, it would be confusing if every phizer product was called phizer.
Phizer makes viagra, the drug has its own name, it would be confusing if every phizer product was called phizer.
Ya. Not surprised. Spin it and spin it. Typical liberal. The confusion is not from the right, sir. Not by a long shot.
But I must say...the left is very devious about deceiving the public. The old "tell a lie often enough and people might believe it" is their motto. It isn't working this time, though.
Sep 27, 2021 08:06:37 #
EmilyD wrote:
Ya. Not surprised. Spin it and spin it. Typical liberal. The confusion is not from the right, sir. Not by a long shot.
But I must say...the left is very devious about deceiving the public. The old "tell a lie often enough and people might believe it" is their motto. It isn't working this time, though.
But I must say...the left is very devious about deceiving the public. The old "tell a lie often enough and people might believe it" is their motto. It isn't working this time, though.
You should look in the mirror. Trump is still claiming that he won the e******n.
Sep 27, 2021 09:22:46 #
moldyoldy wrote:
You should look in the mirror. Trump is still claiming that he won the e******n.
I should look in a mirror because Trump is still claiming that he won??
Okaaaaayyy.......
Sep 27, 2021 09:38:11 #
EmilyD wrote:
I should look in a mirror because Trump is still claiming that he won??
Okaaaaayyy.......
Okaaaaayyy.......
He is still lying.
Sep 27, 2021 10:02:17 #
Sep 27, 2021 10:44:42 #
Sep 27, 2021 10:48:14 #
Sep 28, 2021 17:37:53 #
Michael Rich
Loc: Lapine Oregon
EmilyD wrote:
It's F***i and the other doctors who were advising Trump what to say at the time. Why would they want to shoot down their own advice??
Funny...you think it's okay to post things from CNN, The Daily Beast and factcheck.org.! And you have the nerve to criticize the sources that I deem appropriate? Typical.
Funny...you think it's okay to post things from CNN, The Daily Beast and factcheck.org.! And you have the nerve to criticize the sources that I deem appropriate? Typical.
How many times has F***i been caught lying?
If you want to reply, then register here. Registration is free and your account is created instantly, so you can post right away.
